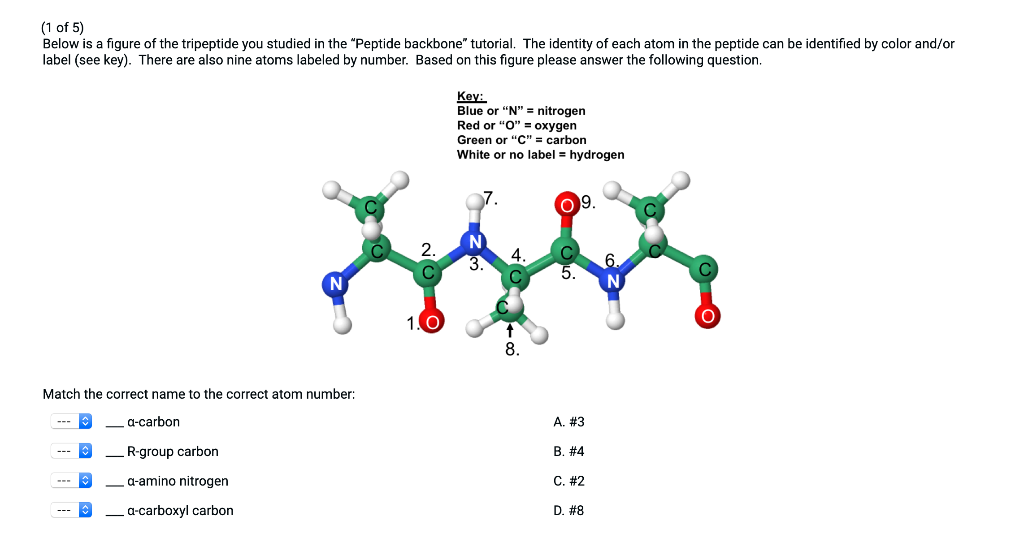
In fact, the term protein sequence is often used interchangeably with primary structure. Primary structure is defined as the linear amino acid sequence of a protein's polypeptide chain. The unit is organized into sections based on both structural and functional relations. Likewise, the PDB‐entry tables given in this unit provide some examples of various folds, but are not comprehensive lists. The scope of this unit is not to enumerate all the existing folds and tertiary structures determined to date, but rather to provide a comprehensive overview of some commonly observed protein fold families and commonly observed structural motifs which have functional significance. Several groups have already attempted to classify protein structures into fold families and superfamiles without focusing on function (Orengo et al., 1993 Murzin et al., 1995). The identification of the fold of a protein has therefore become an invaluable tool since it can potentially provide a direct extrapolation to function, and may allow one to map functionally important regions in the amino acid sequence. As a consequence, structural conservation at the tertiary level is perhaps more profound than it is at the primary. Not only do functionally related proteins generally have similar tertiary structures (see below), but even proteins with very different functions are often found to share the same tertiary folds. The complete structure of a protein can be described at four different levels of complexity: primary, secondary, tertiary, and quaternary structure.Īs a multitude of protein structures are rapidly being determined by X‐ray crystallography and by nuclear magnetic resonance (NMR), it is becoming clear that the number of unique folds is far less than the total number of protein structures. The final 3-D shape of a protein is determined entirely by its primary structure.Proteins fold into stable three‐dimensional shapes, or conformations, that are determined by their amino acid sequence. During a process called dissociation, the polypeptides of a protein with quaternary structure separate and unfold, losing their individual tertiary structure.  
Some diseases may occur because a protein with the correct amino acid sequence fails to correctly fold into its functional form. Cells use chaperones both to accomplish the original folding of some proteins and to restore the structure incorrectly folded ones. Some proteins require other proteins called chaperones in order to correctly fold into their normal 3-D shape. Proteins have a narrow range of conditions in which they fold properly outside the range, proteins tend to renature. 
subsequently returned to its native conditions it will spontaneously refold back to its native structure. If the 3-D structure of a protein depends only on it primary structure and the surrounding environmental conditions, then when the protein is denatured. When normal environmental conditions are reestablished after protein denaturation, almost all proteins can spontaneously refold into their natural shape. Denature proteins are usually biologically active. Proteins can denature when the pH, temp., or ionic concentration of the surrounding solutions change. If a protein's environment is altered, the protein Amy change its shape or even unfold completely, a process called dissociation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
